Pediatric fever of uncertain source: Difference between revisions
Ostermayer (talk | contribs) (Convert 29-60 Days flowchart image to table format matching other sections; keep image as thumbnail) |
|||
| (147 intermediate revisions by 14 users not shown) | |||
| Line 1: | Line 1: | ||
==Background== | ==Background== | ||
*Fever accounts for 30% of pediatric visits | |||
* | *Children <3 months are immunocompromised (e.g. poor opsonization, poor IgG response to encapsulated bacteria, macrophage and neutrophil dysfunction, bone marrow insufficiency) | ||
* | *Concern is for missing a serious bacterial infection (SBI) | ||
* | |||
=== | ===Epidemiology and Risk=== | ||
{| class="wikitable" | |||
| align="center" style="background:#f0f0f0;"|'''Age''' | |||
| align="center" style="background:#f0f0f0;"|'''0-14 days''' | |||
== | | align="center" style="background:#f0f0f0;"|'''14-28 days''' | ||
| align="center" style="background:#f0f0f0;"|'''28-60 days (pre vaccine)''' | |||
| align="center" style="background:#f0f0f0;"|'''28-60 days (post vaccine)''' | |||
| align="center" style="background:#f0f0f0;"|'''60-90 days''' | |||
| align="center" style="background:#f0f0f0;"|'''> 90 days''' | |||
|- | |- | ||
| | | [[Meningitis]]/SBI Prevalence ||1/10||1/20||1/100||1/1000||1/1000-10,000||> 1/10,000 | ||
| | |} | ||
*Serious bacterial illness (SBI) includes UTI, meningitis, pneumonia, bacteremia | |||
*7% of patients <2 years old with fever have [[pneumonia]], however the etiology (viral/bacterial) or even the presence of pneumonia has low inter-observer reliability even among pediatric radiologists<ref>ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545</ref> | |||
*4% Prevalence of [[UTI]] with common other sources of fever ([[OM]], viral [[URI]], etc)<ref>ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545</ref> | |||
0- | *0.3% of previously well children aged 3-36 months who have a fever without a source will develop significant sequelae, 0.03% will develop sepsis or meningitis<ref>ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545</ref> | ||
===Concomitant Respiratory Viral Infection=== | |||
*Relatively high coincidence of [[RSV]], [[enterovirus]], and [[paraflu]] with bacteremia (and UTIs), so positive lab test for these viruses should not change testing and management plan<ref>Greenes, D.S.M., & Harper, M. B.M. (1999). Low risk of bacteremia in febrile children with recognizable viral syndromes. Pediatric Infectious Disease Journal, 18(3), 258-261.</ref> | |||
**[[RSV]]+ neonates aged 0-28 days, 3.7% were bactremic (and 6.1% had [[UTI]]s) | |||
**RSV+ infants aged 29-60 days, (5.5% had [[UTI]]s) | |||
*There is a low coincidence of [[influenza]] with SBI, so postivie lab test for this virus may change testing and management plan (i.e. lower risk of concurrent bacterial illness)<ref>Greenes, D.S.M., & Harper, M. B.M. (1999). Low risk of bacteremia in febrile children with recognizable viral syndromes. Pediatric Infectious Disease Journal, 18(3), 258-261.</ref> | |||
==Clinical Features== | |||
*[[Acute fever|Febrile]] | |||
**Defined as [[Celsius Fahrenheit Temperature Conversion|temperature]] ≥38°C (100.4°F). | |||
**Peripheral temperature is not clinically accurate and central measurements are the preferred means of determining fever. | |||
**Parental report of confirmed fever at home (i.e. via thermometer), even with no fever in ED, has rates of SBIs high as 4.7% (0-28 day range)<ref>Serious bacterial infections in neonates presenting afebrile with history of fever Ramgopal S, Walker LW, Tavarez MM, et al. Pediatrics. 2019;144(2):e20183964.</ref> | |||
==Differential Diagnosis== | |||
{{Pediatric fever DDX}} | |||
==Evaluation & Management== | |||
*Preemies: Count age by estimated postconception date (not by actual delivery date) for 1st-90d | |||
===0-7 Days=== | |||
''For all infants (toxic and well-appearing)'' | |||
{| class="wikitable" | |||
|- | |||
| '''Child Appearance''' | |||
| '''Work Up''' | |||
| '''Treatment''' | |||
| '''Disposition & Follow-up''' | |||
| '''Comments''' | |||
|- | |- | ||
| '''Temperature ≥38°''' | |||
'''Toxic or Well''' | |||
| | | | ||
*CBC | |||
*Blood cultures | |||
*[[Urinalysis]], Urine culture | |||
*[[LP]]-CSF | |||
if | *[[CXR]] | ||
*+/- Stool studies (if diarrhea) | |||
| | | | ||
{{Pediatric fever antibiotics 0-28)}} | |||
| Admit | |||
| SBI incidence | |||
*Ill appearing: 13%–21% | |||
*Not ill appearing: <5% | |||
|} | |||
{{Pediatric fever acyclovir indications}} | |||
Note: | |||
*CXR is optional if no resp sx and another source identified | |||
*LP is necessary even if another source identified due to immature blood-brain barrier | |||
*Do not give ceftriaxone to children <28d as may cause hyperbilirubinemia | |||
===8-21 Days<ref>Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228 </ref>=== | |||
*For toxic infants, treat for sepsis and admit | |||
*For well-appearing infants: | |||
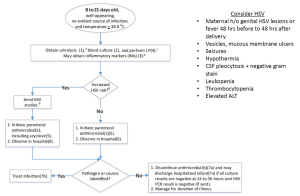
[[File:Peds fever 8-21 days (2.0).png|thumb|8-21 day algorithm]] | |||
{| class="wikitable" | |||
|- | |- | ||
| | | '''Child Appearance''' | ||
| '''Work Up''' | |||
| '''Treatment''' | |||
| '''Disposition & Follow-up''' | |||
| '''Comments''' | |||
| | |||
|- | |- | ||
| | | Temperature ≥38°C | ||
Well-appearing | |||
No evident source of infection | |||
'''Increased HSV risk''' | |||
| | |||
*[[Urinalysis]], Urine culture | |||
*Blood culture | |||
*Inflammatory markers (IMs) - may obtain | |||
*[[LP]]-CSF | |||
*HSV studies | |||
| | |||
*Initiate parenteral antimicrobial(s), including [[Acyclovir]] | |||
| | |||
*Observe in hospital | |||
*If pathogen or source identified → Treat infection | |||
*If all culture results negative at 24-36 hours AND HSV PCR negative → Discontinue antimicrobials; may discharge. Manage for duration of illness. | |||
| | |||
Consider HSV if: | |||
*Maternal h/o genital HSV lesions or fever 48 hrs before to 48 hrs after delivery | |||
*Vesicles, mucous membrane ulcers | |||
*[[Seizures]] | |||
*Hypothermia | |||
*[[CSF]] pleocytosis + negative gram stain | |||
*Leukopenia | |||
*Thrombocytopenia | |||
*Elevated ALT | |||
|- | |- | ||
| | | Temperature ≥38°C | ||
Well-appearing | |||
No evident source of infection | |||
'''No increased HSV risk''' | |||
| | |||
*[[Urinalysis]], Urine culture | |||
*Blood culture | |||
| | *Inflammatory markers (IMs) - may obtain | ||
*[[LP]]-CSF | |||
| | |||
( | *Initiate parenteral antimicrobial(s) | ||
| | |||
*Observe in hospital | |||
*If pathogen or source identified → Treat infection | |||
( | *If all culture results negative at 24-36 hours → Discontinue antimicrobials; may discharge. Manage for duration of illness. | ||
| | |||
| | |||
Treat | |||
| | |||
|} | |} | ||
===22-28 Days<ref>Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228 </ref>=== | |||
*For toxic infants, treat for sepsis and admit | |||
*For well-appearing infants: | |||
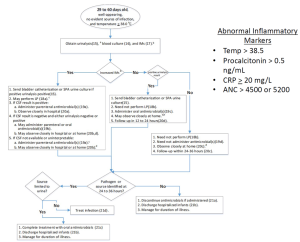
[[File:Peds fever 22-28 days (2.0).PNG|22-28 day algorithm]] | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| '''Child Appearance''' | | '''Child Appearance''' | ||
| '''Work Up''' | | '''Work Up''' | ||
| '''Treatment''' | | '''Treatment''' | ||
| '''Disposition''' | | '''Disposition & Follow-up''' | ||
| '''Follow | | '''Comments''' | ||
|- | |||
| Temperature ≥38°C | |||
Well-appearing | |||
No source of infection | |||
'''Abnormal IMs''' | |||
'''CSF pleocytosis or uninterpretable''' | |||
| | |||
*[[Urinalysis]], Urine culture (bladder catheterization or SPA if UA positive) | |||
*Blood culture | |||
*Inflammatory markers (IMs) | |||
*Perform [[LP]](11b) - CSF | |||
| | |||
*Administer IV antibiotics | |||
| | |||
*Observe in hospital | |||
*If pathogen or source identified → Treat infection | |||
*If all cultures negative at 24-36 hours and HSV PCR negative (if sent) → Discontinue IV antibiotics; may discharge. Follow for duration of illness. | |||
| Abnormal Inflammatory Markers: | |||
*Temp > 38.5 | |||
*[[Procalcitonin]] > 0.5 ng/mL | |||
*CRP ≥ 20 mg/L | |||
*ANC > 4500 or 5200 | |||
|- | |||
| Temperature ≥38°C | |||
Well-appearing | |||
No evident source of infection | |||
'''Abnormal IMs''' | |||
'''No CSF pleocytosis''' (or CSF not obtained) | |||
| | |||
*[[Urinalysis]], Urine culture (bladder catheterization or SPA if UA positive) | |||
*Blood culture | |||
*Inflammatory markers (IMs) | |||
*Perform [[LP]] - CSF (may not be obtained) | |||
| | |||
*If observation at home: Administer IV antibiotics | |||
*If observation in hospital: May administer IV antibiotics | |||
| | |||
*If observation at home: Observe at home. Reassess in 24 hours. | |||
*If observation in hospital: Observe in hospital). | |||
*If pathogen or source identified → Treat infection | |||
*If all cultures negative at 24-36 hours and HSV PCR negative (if sent) → Discontinue IV antibiotics; may discharge. Follow for duration of illness. | |||
| | |||
|- | |||
| Temperature ≥38°C | |||
Well-appearing | |||
No evident source of infection | |||
'''Normal IMs''' | |||
'''LP performed, CSF obtained, CSF pleocytosis or traumatic''' | |||
| | |||
*[[Urinalysis]], Urine culture (bladder catheterization or SPA if UA positive) | |||
*Blood culture | |||
*Inflammatory markers (IMs) | |||
*May perform [[LP]] - CSF | |||
| | |||
*May administer IV antibiotics | |||
| | |||
*Observe in hospital | |||
*If pathogen or source identified → Treat infection | |||
*If all cultures negative at 24-36 hours and HSV PCR negative (if sent) → Discontinue IV antibiotics; may discharge. Follow for duration of illness. | |||
| | |||
|- | |- | ||
| ''' | | Temperature ≥38°C | ||
''' | Well-appearing | ||
No evident source of infection | |||
| | '''Normal IMs''' | ||
'''LP performed, CSF obtained, no CSF pleocytosis/not traumatic''' OR '''CSF not obtained''' OR '''LP not performed''' | |||
| | |||
*[[Urinalysis]], Urine culture (bladder catheterization or SPA if UA positive) | |||
*Blood culture | |||
*Inflammatory markers (IMs) | |||
| | *May perform [[LP]] | ||
| | |||
*If observation at home: Administer IV antibiotics | |||
*If observation in hospital: May administer IV antibiotics | |||
*If LP performed but CSF not obtained, or LP not performed: May administer IV antibiotics); Observe in hospital | |||
| | | | ||
*If observation at home: Observe at home. Reassess in 24 hours. | |||
*If observation in hospital: Observe in hospital. | |||
*If pathogen or source identified → Treat infection | |||
*If all cultures negative at 24-36 hours and HSV PCR negative (if sent) → Discontinue IV antibiotics; may discharge. Follow for duration of illness. | |||
| | |||
|} | |} | ||
===29-60 Days<ref>Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228 </ref>=== | |||
* | *For toxic infants, treat for sepsis and admit | ||
* | *For well-appearing infants: | ||
[[File:Peds fever 29-60 (2.0).PNG|thumb|29-60 day algorithm]] | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| '''Appearance''' | | '''Child Appearance''' | ||
| '''Work Up''' | | '''Work Up''' | ||
| '''Treatment''' | | '''Treatment''' | ||
| '''Disposition''' | | '''Disposition & Follow-up''' | ||
| ''' | | '''Comments''' | ||
|- | |- | ||
| | | Temperature ≥38°C | ||
Well-appearing | |||
No evident source of infection | |||
| | '''Abnormal IMs''' | ||
| | |||
*[[Urinalysis]] (obtain for all) | |||
*Blood culture (obtain for all) | |||
*Inflammatory markers (IMs) (obtain for all) | |||
*Send bladder catheterization or SPA urine culture if positive urinalysis (15) | |||
*May perform [[LP]] (18a) | |||
| | |||
*If CSF result is positive: Administer parenteral antimicrobial(s) (19a) | |||
*If CSF result is negative and either urinalysis negative or positive: May administer parenteral or oral antimicrobial(s) (19b) | |||
*If CSF not available or uninterpretable: Administer parenteral antimicrobial(s) (19a) | |||
| | |||
*If CSF result is positive: Observe closely in hospital (20a) | |||
*If CSF result is negative and either urinalysis negative or positive: May observe closely in hospital or at home (20b,d) | |||
*If CSF not available or uninterpretable: May observe closely in hospital or at home (20b) | |||
| Abnormal Inflammatory Markers: | |||
*Temp > 38.5 | |||
*[[Procalcitonin]] > 0.5 ng/mL | |||
*CRP ≥ 20 mg/L | |||
*ANC > 4500 or 5200 | |||
|- | |- | ||
| | | Temperature ≥38°C | ||
Well-appearing | |||
No evident source of infection | |||
'''Normal IMs''' | |||
'''Positive urinalysis result''' | |||
| | |||
*[[Urinalysis]] (obtain for all) | |||
*Blood culture (obtain for all) | |||
*Inflammatory markers (IMs) (obtain for all) | |||
*Send bladder catheterization or SPA urine culture (15) | |||
*Need not perform [[LP]] (18b) | |||
| | |||
*Administer oral antimicrobial(s) (19c) | |||
| | |||
*May observe closely at home | |||
*Follow-up in 12 to 24 hours (20d) | |||
| | | | ||
|- | |- | ||
| | | Temperature ≥38°C | ||
Well-appearing | |||
No evident source of infection | |||
''' | '''Normal IMs''' | ||
'''Negative urinalysis result''' | |||
| | | | ||
*[[Urinalysis]] (obtain for all) | |||
*Blood culture (obtain for all) | |||
*Inflammatory markers (IMs) (obtain for all) | |||
*Need not perform [[LP]] (18b) | |||
| | |||
| | *Need not administer antimicrobial(s) (19d) | ||
| | |||
*Observe closely at home (20c) | |||
*Follow-up within 24-36 hours (20c) | |||
| | |||
| | |||
|} | |} | ||
'''At 24 to 36 hours follow-up:''' | |||
* | *If pathogen or source identified at 24-36 hours AND source limited to urine → Complete treatment with oral antimicrobials (21c); Discharge hospitalized infants (21b); Manage for duration of illness | ||
*If pathogen or source identified at 24-36 hours AND source NOT limited to urine → Treat infection (21d) | |||
*If NO pathogen or source identified at 24-36 hours → Discontinue antimicrobials if administered (21a); Discharge hospitalized infants (21b); Manage for duration of illness | |||
* | |||
* | |||
===60 Days - 36 Months<ref>Jaskiewicz, J.A., McCarthy, C.A., Richardson, A.C., White, K.C., Fisher, D.J., Powell, K. R., et al. (1994). Febrile infants at low risk for serious bacterial infection-an appraisal of the Rochester criteria and implications for management. Pediatrics 94(3), 390-396.</ref>=== | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| Line 286: | Line 322: | ||
| '''Work Up''' | | '''Work Up''' | ||
| '''Treatment''' | | '''Treatment''' | ||
| '''Disposition | | '''Disposition & Follow-Up''' | ||
|- | |- | ||
| ''' | | '''T≥38° + Toxic''' | ||
| | | | ||
*CBC | |||
*Blood cultures | |||
*[[Urinalysis]], urine culture | |||
*[[LP]]-CSF | |||
*[[CXR]]^ | |||
| | | | ||
{{Pediatric fever antibiotics 90dy-36mo}} | |||
| Admit | | Admit | ||
|- | |- | ||
| '''T≥39°C + Well + Non-complete [[Prevnar]]''' | |||
(No [[Prevnar]] or <4 weeks post 1st [[Prevnar]] dose) | |||
| | | | ||
*[[Urinalysis]], Urine culture | |||
*CBC | |||
*+/- CXR | |||
| If WBC(+): | |||
*[[Ceftriaxone]] 50mg-100mg/kg (also then consider [[blood culture]] and [[LP]], especially in <6mo old) | |||
| Outpatient (24 hour follow-up) | |||
| | |||
[[Ceftriaxone]] 50mg/kg | |||
| Outpatient | |||
|- | |- | ||
| ''' | | '''T≥39°C + Well + [[Prevnar]]''' | ||
(2 [[Prevnar]] or ≥4 weeks post 1st [[Prevnar]] dose) | |||
| | | | ||
*Urine workup (UA, urine culture) for: | |||
**Circumcised males <6 months | **Circumcised males <6 months | ||
**Uncircumcised males <12 months | **Uncircumcised males <12 months | ||
**All females | **All females | ||
*+/- CXR | |||
| Treat [[cystitis]] or [[pneumonia]] if postitive | |||
| Outpatient (48hour follow up) | |||
|- | |||
| '''T≥38-38.9°C + Well''' | |||
| Consider UA, CXR based on symptoms, etc | |||
| Treat [[cystitis]] or [[pneumonia]] if positive | |||
| Outpatient (48-72 hour follow-up)<ref>Baker, M.D., Bello, L.M., & Avner, J.R. (1993). Outpatient management without antibiotics of fever in selected infants. New England Jouranl of Medicine, 329(20), 1437-1441.</ref> | |||
|- | |||
|} | |||
{{Pediatric fever CXR indications}} | |||
*Non-UTI SBI incidence of <.4% in children >6 mo | |||
* | |||
== | ==Low Risk Lab Criteria== | ||
''If low-risk criteria below not met, then perform the LP (if not done) and admit for inpatient antibiotics''<ref> Smitherman, H.F. & Macias, C.G. (2014). Evaluation and management of fever in the neonate and young infant (less than three months of age) [Electronic Version]. UpToDate,Teach, S.J., Kaplan, SL, Wiley, JF.</ref><ref>Dagan, R. Sofer, S., Phillip, M., & Shachak, E. (1988). Ambulatory care of febrile infants younger than 2 months of age classified as being at low risk for having serous bacterial infections. Journal of Pediatrics, 112(3), 355-360.</ref> | |||
==== | ===CBC=== | ||
* | *WBC 5-15 /mm<sup>3</sup> | ||
* | *Absolute Band count <1500 /mm<sup>3</sup> | ||
* | *Procalcitonin ≤0.5 ng/mL | ||
* | *ANC ≤4000/mm<sup>3</sup> | ||
* | ===Urinalysis=== | ||
* | *Clear | ||
==== | *Neg Nitrate and Leukocyte esterase | ||
*WBC <10/high powered field | |||
===CSF=== | |||
*Studies should include WBC, protein, glucose, Gram stain, and culture for bacteria. Consider viral studies (HSV). | |||
====0-28 days==== | |||
*WBC: 0-22/mm<sup>3</sup> | |||
*Protein: <100mg/dL | |||
====>29 days==== | |||
*WBC 0-7/mm<sup>3</sup> | |||
*Protein: 15-25mg/dL | |||
== | ==Additional Management== | ||
===Initial Empiric Antibiotics for Well-Appearing Infants=== | |||
{{Infant fever well antibiotics}} | |||
{{Acetaminophen pediatric dosing chart}} | |||
== | ==See Also== | ||
*[[Acute fever]] | |||
*[[Fever of unknown origin (peds)]] | |||
*[[Urinary tract infection (peds)]] | |||
*[[Sepsis (peds)]] | |||
*[[Meningitis (peds)]] | |||
*[[Febrile seizure]] | |||
*[[PECARN Febrile Infant]] | |||
==External Links== | |||
*[http://www.pemed.org/blog/2011/10/9/fever-of-unknown-source-part-1.html PEM ED Algorithm for Pediatric Fever] | *[http://www.pemed.org/blog/2011/10/9/fever-of-unknown-source-part-1.html PEM ED Algorithm for Pediatric Fever] | ||
*[http://ddxof.com/pediatric-fever/ DDxOf: Pediatric Fever] | |||
== | ==References== | ||
<references/> | |||
[[Category: | [[Category:Pediatrics]] | ||
[[Category:ID]] | |||
Latest revision as of 02:45, 13 April 2026
Background
- Fever accounts for 30% of pediatric visits
- Children <3 months are immunocompromised (e.g. poor opsonization, poor IgG response to encapsulated bacteria, macrophage and neutrophil dysfunction, bone marrow insufficiency)
- Concern is for missing a serious bacterial infection (SBI)
Epidemiology and Risk
| Age | 0-14 days | 14-28 days | 28-60 days (pre vaccine) | 28-60 days (post vaccine) | 60-90 days | > 90 days |
| Meningitis/SBI Prevalence | 1/10 | 1/20 | 1/100 | 1/1000 | 1/1000-10,000 | > 1/10,000 |
- Serious bacterial illness (SBI) includes UTI, meningitis, pneumonia, bacteremia
- 7% of patients <2 years old with fever have pneumonia, however the etiology (viral/bacterial) or even the presence of pneumonia has low inter-observer reliability even among pediatric radiologists[1]
- 4% Prevalence of UTI with common other sources of fever (OM, viral URI, etc)[2]
- 0.3% of previously well children aged 3-36 months who have a fever without a source will develop significant sequelae, 0.03% will develop sepsis or meningitis[3]
Concomitant Respiratory Viral Infection
- Relatively high coincidence of RSV, enterovirus, and paraflu with bacteremia (and UTIs), so positive lab test for these viruses should not change testing and management plan[4]
- There is a low coincidence of influenza with SBI, so postivie lab test for this virus may change testing and management plan (i.e. lower risk of concurrent bacterial illness)[5]
Clinical Features
- Febrile
- Defined as temperature ≥38°C (100.4°F).
- Peripheral temperature is not clinically accurate and central measurements are the preferred means of determining fever.
- Parental report of confirmed fever at home (i.e. via thermometer), even with no fever in ED, has rates of SBIs high as 4.7% (0-28 day range)[6]
Differential Diagnosis
Pediatric fever
- Upper respiratory infection (URI)
- UTI
- Sepsis
- Meningitis
- Febrile seizure
- Juvenile rheumatoid arthritis
- Pneumonia
- Acute otitis media
- Whooping cough
- Unclear source
- Kawasaki disease
- Neonatal HSV
- Specific virus
Evaluation & Management
- Preemies: Count age by estimated postconception date (not by actual delivery date) for 1st-90d
0-7 Days
For all infants (toxic and well-appearing)
| Child Appearance | Work Up | Treatment | Disposition & Follow-up | Comments |
| Temperature ≥38°
Toxic or Well |
|
|
Admit | SBI incidence
|
^Acyclovir if:
Note:
- CXR is optional if no resp sx and another source identified
- LP is necessary even if another source identified due to immature blood-brain barrier
- Do not give ceftriaxone to children <28d as may cause hyperbilirubinemia
8-21 Days[7]
- For toxic infants, treat for sepsis and admit
- For well-appearing infants:
| Child Appearance | Work Up | Treatment | Disposition & Follow-up | Comments |
| Temperature ≥38°C
Well-appearing No evident source of infection Increased HSV risk |
|
|
|
Consider HSV if: |
| Temperature ≥38°C
Well-appearing No evident source of infection No increased HSV risk |
|
|
|
22-28 Days[8]
- For toxic infants, treat for sepsis and admit
- For well-appearing infants:
| Child Appearance | Work Up | Treatment | Disposition & Follow-up | Comments |
| Temperature ≥38°C
Well-appearing No source of infection Abnormal IMs CSF pleocytosis or uninterpretable |
|
|
|
Abnormal Inflammatory Markers:
|
| Temperature ≥38°C
Well-appearing No evident source of infection Abnormal IMs No CSF pleocytosis (or CSF not obtained) |
|
|
|
|
| Temperature ≥38°C
Well-appearing No evident source of infection Normal IMs LP performed, CSF obtained, CSF pleocytosis or traumatic |
|
|
|
|
| Temperature ≥38°C
Well-appearing No evident source of infection Normal IMs LP performed, CSF obtained, no CSF pleocytosis/not traumatic OR CSF not obtained OR LP not performed |
|
|
|
29-60 Days[9]
- For toxic infants, treat for sepsis and admit
- For well-appearing infants:
| Child Appearance | Work Up | Treatment | Disposition & Follow-up | Comments |
| Temperature ≥38°C
Well-appearing No evident source of infection Abnormal IMs |
|
|
|
Abnormal Inflammatory Markers:
|
| Temperature ≥38°C
Well-appearing No evident source of infection Normal IMs Positive urinalysis result |
|
|
|
|
| Temperature ≥38°C
Well-appearing No evident source of infection Normal IMs Negative urinalysis result |
|
|
|
At 24 to 36 hours follow-up:
- If pathogen or source identified at 24-36 hours AND source limited to urine → Complete treatment with oral antimicrobials (21c); Discharge hospitalized infants (21b); Manage for duration of illness
- If pathogen or source identified at 24-36 hours AND source NOT limited to urine → Treat infection (21d)
- If NO pathogen or source identified at 24-36 hours → Discontinue antimicrobials if administered (21a); Discharge hospitalized infants (21b); Manage for duration of illness
60 Days - 36 Months[10]
| Appearance | Work Up | Treatment | Disposition & Follow-Up |
| T≥38° + Toxic |
|
|
Admit |
| T≥39°C + Well + Non-complete Prevnar |
|
If WBC(+):
|
Outpatient (24 hour follow-up) |
| T≥39°C + Well + Prevnar |
|
Treat cystitis or pneumonia if postitive | Outpatient (48hour follow up) |
| T≥38-38.9°C + Well | Consider UA, CXR based on symptoms, etc | Treat cystitis or pneumonia if positive | Outpatient (48-72 hour follow-up)[11] |
- Consider CXR for:
- Respiratory symptoms
- Fever >48 hrs
- Tachypnea
- Hypoxia
- Non-UTI SBI incidence of <.4% in children >6 mo
Low Risk Lab Criteria
If low-risk criteria below not met, then perform the LP (if not done) and admit for inpatient antibiotics[12][13]
CBC
- WBC 5-15 /mm3
- Absolute Band count <1500 /mm3
- Procalcitonin ≤0.5 ng/mL
- ANC ≤4000/mm3
Urinalysis
- Clear
- Neg Nitrate and Leukocyte esterase
- WBC <10/high powered field
CSF
- Studies should include WBC, protein, glucose, Gram stain, and culture for bacteria. Consider viral studies (HSV).
0-28 days
- WBC: 0-22/mm3
- Protein: <100mg/dL
>29 days
- WBC 0-7/mm3
- Protein: 15-25mg/dL
Additional Management
Initial Empiric Antibiotics for Well-Appearing Infants
Neonatal Antibiotics by Source[14]
| Suspected Infection Source | 8-21 Days Old | 22-28 Days Old | 29-60 Days Old |
| UTI |
|
|
|
| No source identified |
|
|
|
| Bacterial meningitis |
|
|
|
Acetaminophen Pediatric Dosing Chart
| Weight (kg) | Weight (lbs) | Age | Dosage (mg) |
| 3-4 | 6-11 | 0-3 mo | 40 |
| 5-7 | 12-17 | 4-11 mo | 80 |
| 8-10 | 18-23 | 1-2 y | 120 |
| 11-15 | 24-35 | 2-3 y | 160 |
| 16-21 | 36-47 | 4-5 y | 240 |
| 22-26 | 48-59 | 6-8 y | 320 |
| 27-32 | 60-71 | 9-10 y | 400 |
| 33-43 | 72-95 | 11 y | 480 |
- Dosage can be given q6 hours
See Also
- Acute fever
- Fever of unknown origin (peds)
- Urinary tract infection (peds)
- Sepsis (peds)
- Meningitis (peds)
- Febrile seizure
- PECARN Febrile Infant
External Links
References
- ↑ ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545
- ↑ ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545
- ↑ ACEP's Clinical Policy on Pediatric Fever. Clinical Policy for Children Younger Than Three Years Presenting to the Emergency Department With Fever. Annuals of Emergency Medicine 2003 42. 530-545
- ↑ Greenes, D.S.M., & Harper, M. B.M. (1999). Low risk of bacteremia in febrile children with recognizable viral syndromes. Pediatric Infectious Disease Journal, 18(3), 258-261.
- ↑ Greenes, D.S.M., & Harper, M. B.M. (1999). Low risk of bacteremia in febrile children with recognizable viral syndromes. Pediatric Infectious Disease Journal, 18(3), 258-261.
- ↑ Serious bacterial infections in neonates presenting afebrile with history of fever Ramgopal S, Walker LW, Tavarez MM, et al. Pediatrics. 2019;144(2):e20183964.
- ↑ Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228
- ↑ Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228
- ↑ Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228
- ↑ Jaskiewicz, J.A., McCarthy, C.A., Richardson, A.C., White, K.C., Fisher, D.J., Powell, K. R., et al. (1994). Febrile infants at low risk for serious bacterial infection-an appraisal of the Rochester criteria and implications for management. Pediatrics 94(3), 390-396.
- ↑ Baker, M.D., Bello, L.M., & Avner, J.R. (1993). Outpatient management without antibiotics of fever in selected infants. New England Jouranl of Medicine, 329(20), 1437-1441.
- ↑ Smitherman, H.F. & Macias, C.G. (2014). Evaluation and management of fever in the neonate and young infant (less than three months of age) [Electronic Version]. UpToDate,Teach, S.J., Kaplan, SL, Wiley, JF.
- ↑ Dagan, R. Sofer, S., Phillip, M., & Shachak, E. (1988). Ambulatory care of febrile infants younger than 2 months of age classified as being at low risk for having serous bacterial infections. Journal of Pediatrics, 112(3), 355-360.
- ↑ Evaluation and Management of Well-Appearing Febrile Infants 8 to 60 Days Old Robert H. Pantell, Kenneth B. Roberts, William G. Adams, Benard P. Dreyer, Nathan Kuppermann, Sean T. O'Leary, Kymika Okechukwu and Charles R. Woods; Subcommittee On Febrile Infants Pediatrics July 2021, e2021052228; DOI: https://doi.org/10.1542/peds.2021-052228